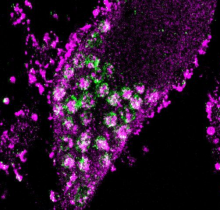
In a new paper published in Nature Neuroscience, the Kampmann lab conducted the first genome-wide CRISPR-based screens in human neurons, and uncovered a new pathway protecting neurons from oxidative stress.
Surprisingly, the screens found a neuron-specific role for the lysosomal protein prosaposin in controlling survival during oxidative stress. Prosaposin loss caused the formation of lipofuscin, a hallmark of aging, which trapped iron, generating ROS and triggering ferroptosis. Mutations in prosaposin have been linked to neurodegenerative diseases.
The project was led by Ruilin Tian, a graduate student in the Kampmann, who recently started his own lab as an Assistant Professor at the Southern University of Science and Technology in Shenzhen, China.
The Kampmann lab collaborated with Data Tecnica International to create a data commons, called CRISPRbrain, to enable the scientific community to access, explore, visualize and cross-compare the results of CRISPR screens in different human cell types.
Read the UCSF press release here.
Read a discussion of this research by the Alzheimer’s Forum here.
Reference:
Tian R, Abarientos A, Hong J, Hashemi SH, Yan R, Dräger N, Leng K, Nalls MA, Singleton AB, Xu K, Faghri F, Kampmann M.
Genome-wide CRISPRi/a screens in human neurons link lysosomal failure to ferroptosis.
Nature Neuroscience. 2021 May 24. doi: 10.1038/s41593-021-00862-0. Epub ahead of print. PMID: 34031600.
Access the preprint on bioRxiv here.